Development and Validation of a simple UV spectrophotometric method for the determination of Delamanid
Abstract
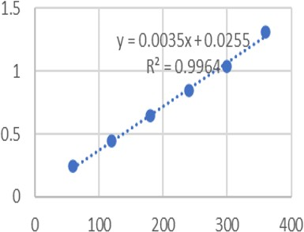
Using a Shimadzu UV-2600, a quick, precise, easy, and affordable UV spectrophotometric approach has been created. Solvent made with methanol to assess the bulk Delamanid content. A wavelength of 320 nm was used for the detection process. The parameters linearity, accuracy, precision, ruggedness, robustness, LOD, and LOQ were taken into consideration during method validation in accordance with ICH Q2R1 criteria. It demonstrated linearity in the range of 60-360 (/mL) at a predetermined λmax of 320 nm, and it had a strong correlation coefficient (R2-0.996) and outstanding mean recovery (99.00-100.07%). Determination of Delamanid used this technique effectively. The method's linearity, accuracy, repeatability, and reproducibility were statistically and by recovery experiments confirmed. The outcomes demonstrated the method's applicability for both regular Delamanid bulk analysis and commercial formulations.
Full text article
References
A Bahuguna and D S Rawat. An overview of new antitubercular drugs, drug candidates, and their targets. Medicinal Research Reviews, 40(1):263–292, 2020.
S K Field, D Fisher, J M Jarand, and R L Cowie. New treatment options for multidrug-resistant tuberculosis. Ther Adv Respir Dis, 6(5):255–268, 2012.
M Matsumoto, H Hashizume, and T Tomishige. OPC-67683, a nitro-dihydro-imidazooxazole derivative with promising action against tuberculosis in vitro and mice. PLOS Med, 3(11):2131–2144, 2006.
Validation of analytical procedure: text and methodology. International Conference on Harmonizations, pages 1–17, 2005.
M S Glickman and W R Jacobs. Microbial pathogenesis of Mycobacterium tuberculosis: dawn of a discipline. Cell, 104(4):236–243, 2001.
Stephen K Field. Safety and Efficacy of Delamanid in the Treatment of Multidrug-Resistant Tuberculosis. Clinical medicine insight: therapeutics, 5(5):137–149, 2013.
J Nicola, Jin Han Ryan, and Lo. Delamanid: first global approval. Drugs, 74(9):1041–1045, 2014.
Authors

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.