Abstract
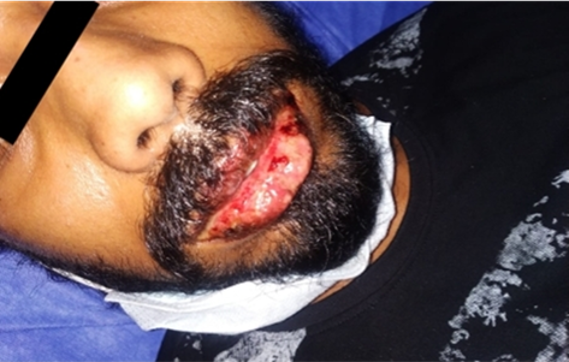
Stevens-Johnson Syndrome (SJS) is an acute, self-limited, rare but life threatening disease that manifests as severe mucocutaneous blistering and erosions. Here we report a rare case of allopurinol-induced SJS. A 25-year-old male patient with no other comorbidities was admitted to the hospital with complaints of fever, redness of eyes, swelling of lips with discharge and crusting, extensive erosions in the oral mucosa for the last 4 days, following consumption of allopurinol for a duration of 1 month. Investigations were within normal limits. The offending drug was withdrawn and he was treated with corticosteroids, antimicrobials, and other supportive measures. Allopurinol, a Xanthine oxidase inhibitor is mostly used for the treatment of primary and secondary hyperuricemia, Health care professionals must be aware of the spectrum of adverse effects of this drug and must take urgent measures once the diagnosis is suspected especially to save the patient from such severe or fatal reactions like SJS/Toxic epidermal necrolysis (TEN).
Full text article
References
Bastuji-Garin, S. 1993. Clinical Classification of Cases of Toxic Epidermal Necrolysis, Stevens- Johnson Syndrome, and Erythema Multiforme. Archives of Dermatology, 129(1):92–96.
Beveridge, J., Harris, M., Wise, G., Stevens, L. 1964. Long-Acting Sulphonamides Associated with Stevens-Johnson Syndrome. The Lancet, 284(7359):593–593.
Chan, H. L. 1990. The incidence of erythema multiforme, Stevens-Johnson syndrome, and toxic epidermal necrolysis. A population-based study with particular reference to reactions caused by drugs among outpatients. Archives of Dermatology, 126(1):43–47.
Guitart, J. 1995. Immunopathology of Stevens- Johnson Syndrome. Allergy and Asthma Proceedings, 16(4):163–164.
Martínez-Cabriales, S., Martínez-Cabriales, A. 2015. Review of News in severe clinical adverse drug reactions: Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN). Gac Med Mex, 151(6):777–787.
Roujeau, J.-C. 1987. Genetic Susceptibility to Toxic Epidermal Necrolysis. Archives of Dermatology, 123(9):1171–1173.
Roujeau, J. C. 1996. Medication Use and the Risk of Stevens-Johnson Syndrome or Toxic Epidermal Necrolysis. American Journal of Ophthalmology, 121(3):341–341.
Roujeau, J.-C., Chosidow, O., Saiag, P., Guillaume, J.C. 1990. Toxic epidermal necrolysis (Lyell syndrome). Journal of the American Academy of Dermatology, 23(6):1039–1058.
Thiers, B. H. 2006. HLA-B*5801 Allele as a Genetic Marker for Severe Cutaneous Adverse Reactions Caused by Allopurinol. Yearbook of Dermatology and Dermatologic Surgery, pages 212–213.
Authors

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.